|
A conductivity meter is used to measure the conductivities of electrolytic solutions. Electrodes on the device are dipped into a solution, and the solution's electrical conductivity is registered on the display. Electrical conductivity is defined as the transfer of an electric current through a solid or liquid. In electrolytic solutions, the current is carried by ions, as in solutions of salts, acids or bases. Conductivity is inversely proportional to the resistivity of the solution.
Electrolytes are compounds that produce ions when dissolved in water. Because charged ions are present and can move from place to place, solutions of electrolytes conduct electricity.
Solutions of strong electrolytes are good conductors of electricity, which indicates that many ions are present. A strong electrolyte is converted completely to ions in water. Examples are: NaCl, HCl, and NaOH.
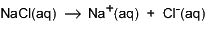
Solutions of weak electrolytes, such as ammonia and acetic acid, are poor conductors, which indicates that they are only partially converted to ions in water. Some ammonia or acetic acid molecules are still present.
Non-electolytes such as alcohol or sugar do not produce ions in water, so solutions of these substances do not conduct electricity.
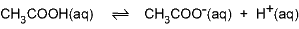
A substance that consists of ions, such as NaCl, is said to disassociate when dissolved in water. This means that its ions separate and can move nearly independently of each other among the water molecules.
A molecular substance, such as hydrogen chloride or ammonia, is said to ionize when dissolved in water. This means that the molecules react with water to form ions, but the ions were not present until the substance came into contact with water.
|