
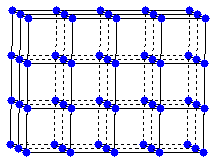
One description of the cesium chloride structure places chloride ions on the lattice point of a simple cubic space lattice.
The cesium ions occupy all of the cubic holes in the simple cubic array of chloride ions.
The unit cell of cesium chloride is a cube with a lattice point at each corner. Chloride ions are located on the corners and a cesium ion at the center of the most common cell.
Click the links on the right to illustrate these topics further. The underlined links above go to definitions.
A
simple cubic lattice

A
cubic array of chloride ions
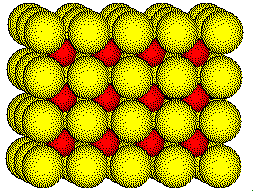
Cesium
ions occupy cubic holes
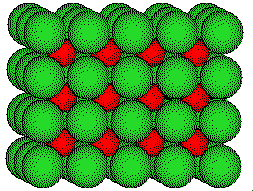

The
CsCl unit cell
Copyright ©
2003
by: Journal of Chemical Education Software
by: Journal of Chemical Education Software